Evolutionarily conserved protein motifs drive interactions between the plant nucleoskeleton and nuclear pores
Auteurs :Mermet S , Voisin M , Mordier J , Dubos T , Tutois S , Tuffery P, Baroux C, Tamura K, Probst AV , Vanrobays E , Tatout C
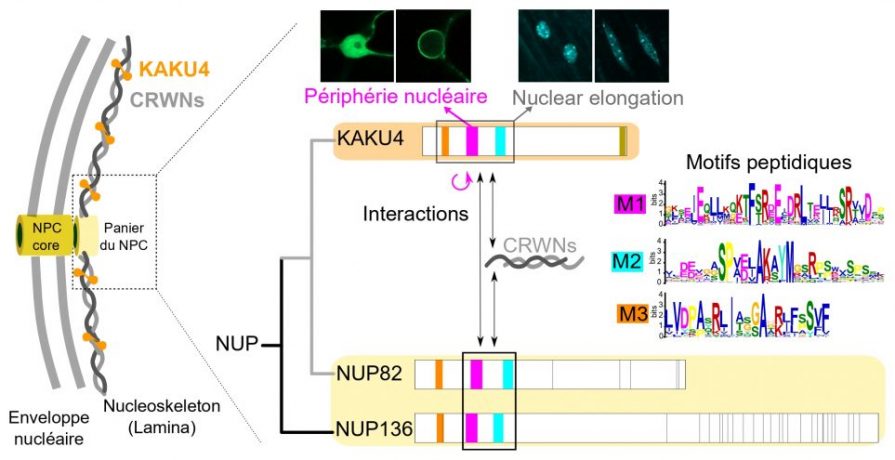
The nuclear envelope is made up of a double membrane that separates the nucleus from the cytoplasm. It isolates and protects the DNA and creates two functional compartments with DNA replication and transcription taking place inside the nucleus and translation in the cytoplasmic compartment. The nuclear envelope is associated inside the nucleus with the nucleoskeleton, a nuclear structure constituted of lamins and their associated proteins. It is punctuated by thousands of nuclear pore complexes (NPCs) that not only regulate the trafficking of macromolecules into and out of the nucleus but also play significant roles in transcription regulation.
To function in a coordinated manner, the nuclear envelope, NPCs and nucleoskeleton must be closely linked by a network of protein-protein interactions. These are poorly documented in plants as the plant-specific proteins within these structures have only recently been described. This motivated us to investigate the characteristics and functions of these interactions.
One such plant-specific protein is KAKU4, a component of the nucleoskeleton in which, using Arabidopsis as a model species, we identified three short peptide motifs of about 30 amino acids each. These motifs allow protein protein interaction with the CRWN proteins, the main component of the plant nucleoskeleton and are required for elongation of the nucleus in differentiating tissues. The motifs were also found in NUP82 and NUP136, two plant-specific nucleoporins, suggesting that KAKU4 diverged from an ancestral nucleoporin, which had the ability to interact with the nucleoskeleton, and led to the emergence of KAKU4 as a new component of the plant nucleoskeleton.
In future, it will be important to assess if the NPC-nucleoskeleton physical interactions identified are essential to anchor the nucleoskeleton at the nuclear envelope and to control NPC composition and distribution. Whether they regulate gene expression or chromatin organisation in response to stress should also be investigated.
dans The Plant Cell